Avivagen Inc. (TSXV:VIV, OTCQB:VIVXF), makers of oxy-beta carotene compounds (OxC-beta) that when added to feedstock make it healthier for animals by boosting their natural immunity to illness and disease, recently secured a 1.2-tonne order from AB Vista, a U.K.-based subsidiary of Associated British Foods Plc (ABF:LSE), which employs some 137,000 people in 50 countries and posted roughly £15.6 billion (US$19 billion) in sales last year.
AB Vista plans to use OxC-beta Livestock in Brazil where the U.K. company is running trials with several large poultry and cattle producers.
Brazil is ranked as third-largest feed-producing country in the world and AB Vista is the exclusive distribution partner for OxC-beta in Brazil, Thailand and the United States.
Avivagen boasts that numerous international livestock trials using OxC-beta Livestock, mostly with pigs and chickens, have proven that the product performs as well as, and sometimes better than in-feed antibiotics.
"We believe (Avivagen) is at an inflection point with significant sales increases just around the corner and presents a favorable risk/reward proposition." — Senior Research Analyst Joe Gomes of Noble Capital Markets
The company hopes to one day replace all in-feed antibiotics as growth promoters.
“The large size of this order is a testament to the growing demand for safe and effective alternatives to antibiotics, and the market opportunity for our products," said Avivagen CEO Kym Anthony.
OxC-beta Livestock is currently available for sale in the United States, Philippines, Mexico, Taiwan, New Zealand, Thailand, Brazil, Australia, Vietnam and Malaysia.
Technical Analyst Clive Maund, seizing upon the size of the new feedstock order, wrote that demand for Avivagen’s product is improving because “management has been cleaned up with positive changes and the company is implementing a Smart marketing program and all this together is making it an attractive takeover candidate, given its current low valuation.”
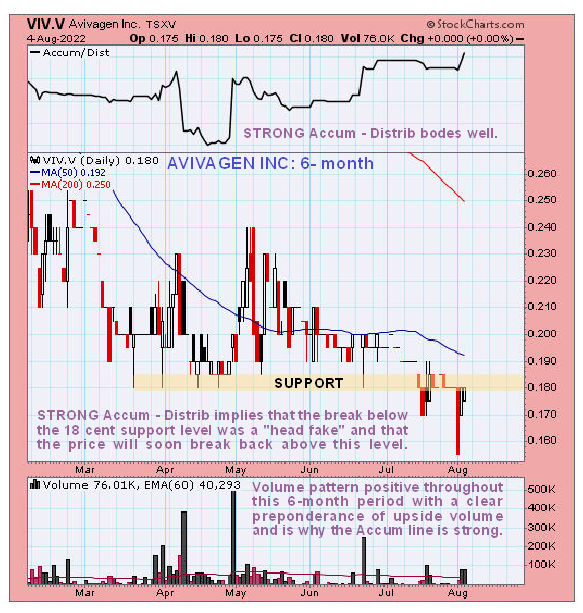
He notes that Avivagen, at its peak in 2007, traded at about 20 times the new lows set this year but the healthy accumulation line on Maund’s technical chart implies that a reversal is indeed incubating.
Maund calls Avivagen “a strong buy here for all timeframes on both fundamental and technical grounds” and recommends that investors “stay long” and add to positions on the dips. Avivagen closed at CA$0.18 on August 10, 2022.
Over at Chicago-based Zack’s Small-cap Research, Senior Analyst David Bautz in a May 9, 2022 research report places a valuation of CA$1.25 on Avivagen, while Joe Gomes, senior research analyst with Boca Raton, Florida-based Noble Capital Markets, gives Avivagen a 12-month target of US$1 and an Outperform rating.
Gomes wrote, “We believe (Avivagen) is at an inflection point with significant sales increases just around the corner and presents a favorable risk/reward proposition.”
New Sales Orders
The sales order with AB Vista comes on the heels of a 1,500-unit order for Vivamune from Avivagen’s distribution partner in Taiwan — it’s the largest order to date from the partnership.
The company’s OxC-beta complex is a key ingredient in canned food and kibble made by a premium dog food maker in Taiwan.
In February 2022, Avivagen said that OxC-beta was approved for use in Vietnam, which is the largest feed market in Southeast Asia. Total feed consumption in Vietnam was 32 million tonnes in 2020, and it is expected to grow to 35 million tonnes in the next few years.
Meanwhile, Avivagen has booked two other orders in Mexico.
The milk from cattle consuming Oxy-beta Dairy had 139.78 colony forming units (bacteria cells) per milliliter (CFU/ml), while the cows without had 657 CFU/ml — that’s 370% less bacteria.
A distributor of high-quality nutritional products for dogs ordered 4,000 units of “white label” product that it plans to market under its own brand, while another Mexican customer ordered 500 units of Vivamune (following a successful internal study), which it plans to market through veterinarians and hospitals.
The Mexican orders come about three months after the company secured an order from a large poultry producer in Mexico.
The Business
Avivagen makes money by selling its patented OxC-beta formulas — basically fully-oxidized beta-carotene — to companies that make livestock feed, vitamins and other products for human consumption, as well as pet food — but the fastest path to growth will be to find a way into the U.S. feedstock market, which today is worth roughly US$1.2 billion annually.
The pet food market is worth about US$635 million annually but the biggest market by far is that of human consumption — worth some US$90 billion every year.
So far, Avivagen’s only human product offering is the Dr. Tobias Beta Blend, a beta carotene supplement available on Amazon, but the company has yet to get its No Objection Letter from the U.S. Food and Drug Administration, which would give it carte blanche in the U.S. market.
“The big picture is the human market, but I think the market that's in front of us and the market that we'll be able to capitalize on, is agricultural fields,” Independent investor Penny Queen told Streetwise Reports.
The most likely path to the U.S. feedstock market is also through AB Vista, which is Avivagen’s U.S. partner. AB Vista already has relationships with ADM and Cargill, the two major U.S.-based agribusinesses.
Last year, Avivagen sold about 10 tonnes of product to AB Vista, but the plan is to boost that to 500 tonnes annually. The recent Brazilian deal puts Avivagen right on track to meet and even exceed those goals.
The company produces its product through Allied Biotech, a beta carotene manufacturer in Taiwan and the third-largest producer of beta carotene in the world, behind only German firm BASF and Dutch company DSM. Jolyon Burton, a large shareholder in Avivagen, suggests that if sales increase dramatically, as expected, it makes sense to build another manufacturing facility in the American Midwest to supply the Americas. He estimates it would cost around US$5 million.
A Recent Study...
Ottawa-based Avivagen recently published the results of a study on the positive effects of one of the company’s most promising products, OxC-beta Dairy.
Dairy cattle in China suffering from either clinical or subclinical mastitis (an internal inflammation that causes visibly abnormal milk) were divided into two groups: one herd was given OxC-beta augmented feed, while the other group consumed its usual diet.
The cows were given 3 grams of OxC-beta Dairy per day over 35 days. The milk from cattle consuming Oxy-beta Dairy had 139.78 colony forming units (bacteria cells) per milliliter (CFU/ml), while the cows without had 657 CFU/ml — that’s 370% less bacteria.
The OxC-Beta Dairy test group also delivered more milk fat (4.56% vs 3.83%), as well as more protein (3.82% vs 3.3%). Perhaps more importantly, other similar studies have shown that Oxy-beta Dairy contributed to as much as 9.7% more milk per day.
“With these data, new customers and new contracts are imminent. For instance, we are negotiating with the Dairy Association of Mexico,” said Burton, who suggested that the deal could start with a couple of tonnes of OxC-beta per month.
Avivagen says the Chinese dairy study supports its claims that OxC-beta Dairy possesses “immune optimizing activities” that help animals resist infections and limit excessive levels of inflammation. Basically, it means the animals are far more likely to remain healthy during periods of stress.
But what’s in it? OxC-Beta Dairy contains synthesized polymer compounds that occur naturally in certain plants. Many people think of plastics when they think of polymers but in scientific terms a polymer is a class of natural or man-made substances composed of large molecules, known as macromolecules, which are multiples of simpler chemical units called monomers.
What does it all mean? Essentially, it means that Avivagen developed a method to make these naturally occurring oxidized beta carotene polymers — natural stuff that is man-made — and the process is not only patented but meets the standards set out in FAMIqs, a global quality and feed safety management system for specialty feed ingredients.
“It does occur in nature, but in minute quantities and not enough to get to get the utility out. So the manufacturing process guarantees the purity and quantity. But it is something that exists in nature,” Burton explained to Streetwise Reports.
Burton adds that Avivagen will own the intellectual property rights on its OxC-beta technology until the mid-2030s.
It was developed in the 1990s in partnership with Canada’s National Research Council in Ottawa. Rights to the technology were put into a shell company that went public in 2005.
Avivagen has about 73 million shares outstanding, most of which is free floating. It has no significant institutional ownership, and it trades in a 52-week range CA$0.32 and CA$0.18.
| Want to be the first to know about interesting Biotechnology / Pharmaceuticals investment ideas? Sign up to receive the FREE Streetwise Reports' newsletter. | Subscribe |
Disclosures:
1) Brian Sylvester wrote this article for Streetwise Reports LLC and provides services to Streetwise Reports as an independent contractor. He or members of his household own securities of the following companies mentioned in the article: None. He and members of his household are paid by the following companies mentioned in this article: None. His company has a financial relationship with the following companies referred to in this article: None.
2) The following companies mentioned in this article are billboard sponsors of Streetwise Reports: None. Click here for important disclosures about sponsor fees. The information provided above is for informational purposes only and is not a recommendation to buy or sell any security. As of the date of this article, an affiliate of Streetwise Reports has a consulting relationship with Avivagen Inc. Please click here for more information.
3) The article does not constitute investment advice. Each reader is encouraged to consult with his or her individual financial professional and any action a reader takes as a result of information presented here is his or her own responsibility. By opening this page, each reader accepts and agrees to Streetwise Reports' terms of use and full legal disclaimer. This article is not a solicitation for investment. Streetwise Reports does not render general or specific investment advice and the information on Streetwise Reports should not be considered a recommendation to buy or sell any security. Streetwise Reports does not endorse or recommend the business, products, services or securities of any company mentioned on Streetwise Reports.
4) From time to time, Streetwise Reports LLC and its directors, officers, employees or members of their families, as well as persons interviewed for articles and interviews on the site, may have a long or short position in securities mentioned. Directors, officers, employees or members of their immediate families are prohibited from making purchases and/or sales of those securities in the open market or otherwise from the time of the decision to publish an article until three business days after the publication of the article. The foregoing prohibition does not apply to articles that in substance only restate previously published company releases. As of the date of this article, officers and/or employees of Streetwise Reports LLC (including members of their household) own securities of Avivagen Inc., a company mentioned in this article.

















































